|
Paper II illustrates how findings from this research informed the development of a tutorial that led to significant improvement in student understanding of atomic emission spectra. This article (Paper I) describes how several serious conceptual and reasoning difficulties were identified among students as they tried to relate a discrete line spectrum to the energy levels of atoms in a light source. The photon is emitted with the electron moving from a higher energy level. If a hydrogen atom could have any value of energy, then a continuous spectrum would have been observed, similar to blackbody radiation. Bohr could now precisely describe the processes of absorption and emission in terms of electronic structure. Thus the energy levels of a hydrogen atom had to be quantized in other words, only states that had certain values of energy were possible, or allowed. 
About half of the latter group were preservice high school physics teachers. The energy of an emitted/absorbed photon is set by the difference in energy levels for a jump from one energy level with a given principle quantum number nlower. When the electron changes levels, it decreases energy and the atom emits photons. The Balmer seriesthe spectral lines in the visible region of hydrogen's emission spectrumcorresponds to electrons relaxing from n3-6 energy levels to the n2 energy level. The others included graduate and undergraduate teaching assistants at UW and physics majors in introductory and advanced physics courses at the University of Zagreb, Zagreb, Croatia. Most of the more than 1000 students who participated in this four-year investigation were science majors enrolled in the introductory calculus-based physics course at the University of Washington (UW) in Seattle, WA, USA. mostly all textbooks on the spectral lines of elements tend to only go up to uranium, however, textbooks go into much more detail for each element.This is the first of two closely related articles (Paper I and Paper II) that together illustrate how research in physics education has helped guide the design of instruction that has proved effective in improving student understanding of atomic spectroscopy. When the electron falls back to the lower energy level, light is. You can see a list of of all the known spectral lines of elements on Wikipedia since it seems to have the most updated table. When an atom or molecule absorbs energy, the electrons are excited to a higher energy level. Well, only the photons that have a wavelength. These heavier elements likely have their own spectral lines, however, due to all I stated above, it isn't exactly possible to measure. What will happen if you shine a spectrum of light of different wavelengths (continuous spectrum) on the atom. 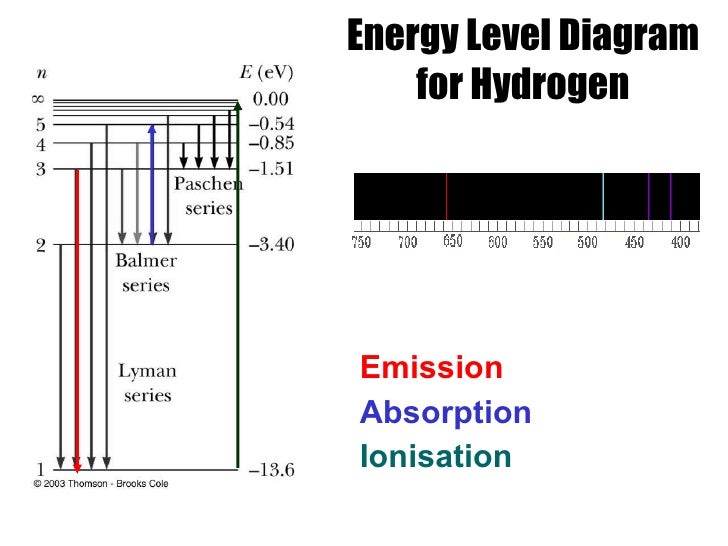
This isn't even considering how much this would cost. To return to ground states from the excited state, which. This would make measuring their spectral lines nearly impossible. When an atoms electrons absorb energy, they migrate from lower energy levels to higher energy levels. It's incredible because some of the heavier and more unstable elements have insanely short half lives ranging from 100.5 days (the most stable isotope of fermium (Fm, 100)), to 0.69 microseconds (0.00069 milliseconds) (Oganesson (Og, 118)). a spectroscope that are emitted by electrons as they transition from higher to lower energy levels unique for each element. Kirchhoffs Laws Thus, emission spectra are produced by thin gases in which the atoms do not experience many collisions (because of the low density). I could not find any data as to why this may be, however, I believe it is simply because we could not test for the spectral lines of the heavier elements due to their instability and scarcity. Emission Spectrum Whenever electromagnetic radiation interacts with atoms and molecules of matter, the electrons in these atoms may absorb energy and jump to a. Also, tables of the spectral lines of elements only seem to go up to the 99th element, Einsteinium (not including Astatine (At, 85) and Francium (Fr, 87). In summary, emission spectra are produced when atoms emit photons as their electrons transition from excited states to lower energy levels. However, it is much more difficult to observe the spectral lines of liquids and solids due to how close together the atoms are. Each element has its own distinct spectral line and this fact can and has been used to identify an element. 
Actually, this phenomena can be observed with liquids and solids as well. Bohr became convinced of its validity and spent part of 1912 at Rutherford’s laboratory.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
